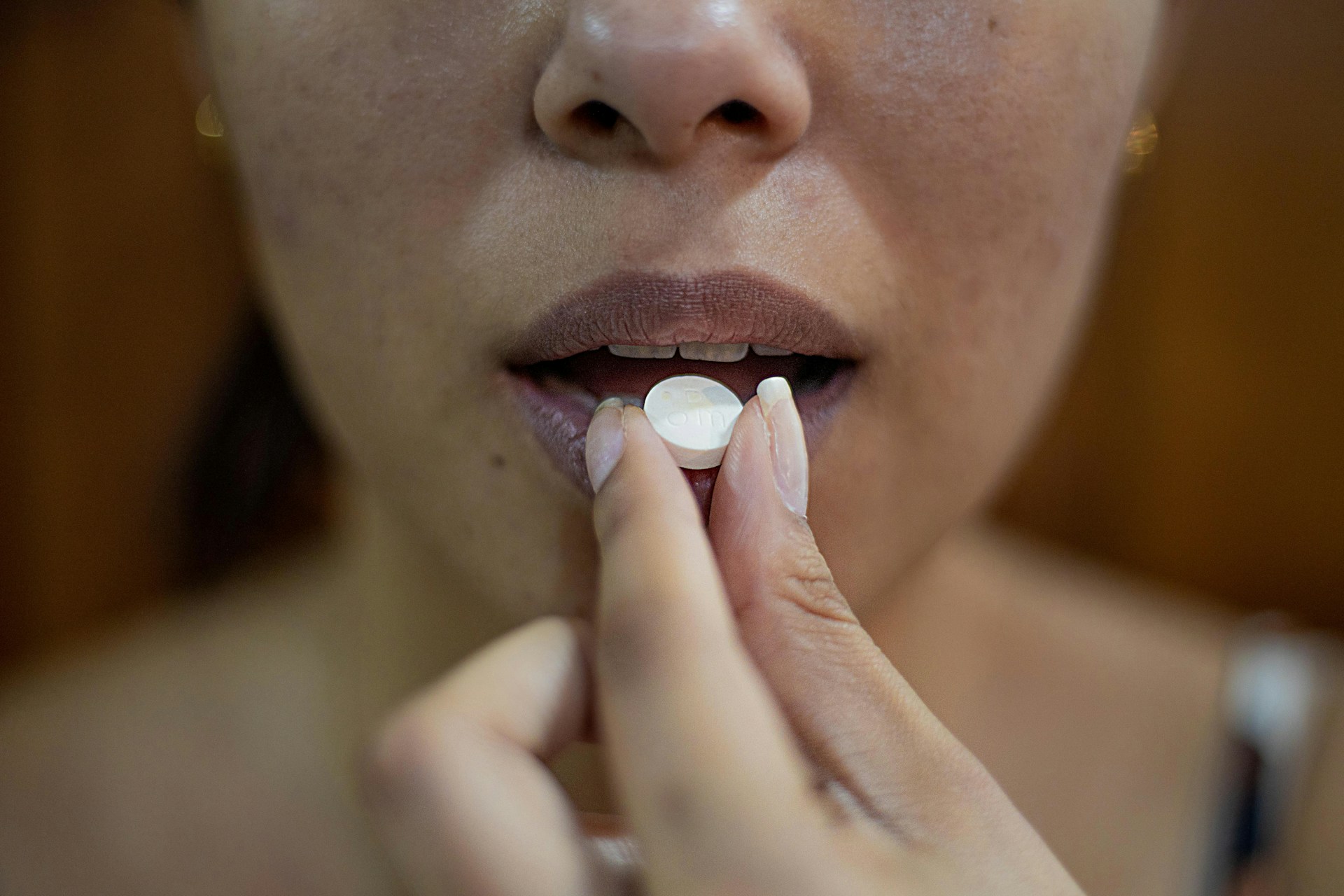
The 5th Circuit Court of Appeals has temporarily blocked a Biden-era policy that permitted the mailing of abortion drugs without in-person doctor consultations, following a unanimous ruling by a three-judge panel on Friday. This decision underscores ongoing tensions surrounding abortion access and safety regulations in the United States.
Explainer Trump Administration Reassesses Approach to Sanctuary Officials
The court's ruling comes amid concerns that the relaxed regulations could lead to increased health risks for women and unauthorized abortions. Louisiana's legal challenge highlighted significant issues with the FDA's data supporting the policy change, claiming it resulted in numerous illegal abortions and financial burdens on the state’s Medicaid system.
Mifepristone, the primary drug used in medical abortions, was first approved by the U.S. Food and Drug Administration (FDA) in 2000 with strict safety protocols requiring in-person evaluations. These regulations were designed to protect women from potential complications, such as ectopic pregnancies, which can be life-threatening if treated improperly. However, in 2023, the FDA, under the Biden administration, lifted these safeguards, allowing abortion pills to be mailed without any in-person checks.
Key Details
In 2025, Louisiana filed a lawsuit against the FDA, arguing that the agency's decision to relax safety measures was not supported by adequate data. The state reported that the new policy led to approximately 1,000 illegal abortions each month and increased Medicaid costs due to complications arising from the use of mifepristone. The 5th Circuit noted that Louisiana had demonstrated standing and was likely to succeed in its case, thus warranting a stay on the policy.
Background and Reactions
Judge Kyle Duncan, writing for the panel, stated, "We have now three times found that the agency’s progressive relaxation of mifepristone’s guardrails likely lacked a basis in data and scientific literature." The court's decision emphasized the potential harm to women and the inadequacy of the FDA's previous assessments.
The FDA's acknowledgment of procedural flaws in its decision-making process was highlighted in a letter from Health and Human Services Secretary Robert F. Kennedy Jr. and FDA Commissioner Marty Makary in September 2025. They admitted to receiving reports of serious adverse events related to mifepristone and recognized a "lack of adequate consideration" in prior approvals. This admission came after 22 state attorneys general urged the FDA to reconsider the mail-order policy.
A study conducted by the Ethics and Public Policy Center revealed alarming statistics regarding the safety of mifepristone. It found that 10.93 percent of women experienced severe complications, such as sepsis or hemorrhaging, within 45 days of taking the drug. This rate is significantly higher than the less than 0.5 percent reported during clinical trials, raising further concerns about the drug's safety profile.
In light of these findings, over 100 organizations have called for the reinstatement of safety measures for mifepristone. A poll from August 2025 indicated that even among those who support abortion rights, there are significant concerns regarding the safety of the drug and a desire for restored protections.
Despite the mounting evidence and public concern, the FDA has been criticized for its slow response to safety issues related to mifepristone. Pro-life advocates argue that the agency's inaction has forced states like Louisiana to seek legal remedies. Marjorie Dannenfelser, president of SBA Pro-Life America, stated, "It’s shameful that the Trump administration’s inaction has forced pro-life states to take their battle to the federal courts. The FDA does not need a year or more to complete a comprehensive study before it can take dangerous abortion drugs out of the mail."
The FDA's current stance appears to be one of caution, as it has requested that the district court pause Louisiana's lawsuit pending the completion of its internal review of mifepristone. This decision has left many advocates on both sides of the abortion debate awaiting further developments.
As the legal battle continues, the implications of the 5th Circuit's ruling could have lasting effects on abortion access and safety regulations across the country. The court's decision reflects a growing scrutiny of federal policies that impact women's health and reproductive rights, setting the stage for further legal and political challenges ahead.
Why it matters
- Referenced surveys and datasets are best read as descriptive and correlational unless the underlying research clearly establishes causation.
- The story shows how legal and policy fights move from proposals and hearings into concrete consequences for institutions and families.
- The story highlights how struggles over policy and power inside institutions end up shaping daily life for ordinary people.
What’s next
- Watch for the next formal step mentioned in the story, such as a committee hearing, court date, rulemaking notice, or floor vote.
- Readers can follow the agencies, lawmakers, courts, or organizations cited here to see how their decisions evolve after this story.
- Subsequent filings, rulings, votes, or agency announcements may clarify how durable these changes prove to be over time.